In order to provide the best possible care to patients with a rare condition, it is essential that global knowledge about the condition is gathered. Nationwide, centers of expertise have been set up to stimulate care for rare disorders and to gather knowledge. For the formal recognition of an expertise center by the Ministry of Health, an important condition is that the expertise center gathers, analyzes and shares knowledge through publications. These can be publications in scientific journals, but also treatment guidelines for health care professionals or information brochures for patients or caregivers. We optimize care and research within ENCORE through standardized follow-up and close collaboration between doctors and researchers. That way, we can ultimately develop better treatments for rare conditions. You may therefore be asked to participate in research. Participation in research is always on a voluntary basis. The data obtained is stored and analyzed in an anonymous form. All research has been approved in advance by an ethics review committee.
Genetic testing will be performed on all NF1 patients seen in our center of expertise to determine the genetic cause and to be able to support and advise the parents. If genetic testing has already been done elsewhere, it will not be repeated. This genetic knowledge also helps us to better understand the effect of the genetic change ("mutation") on the severity of symptoms. We can then also investigate which treatment works best for a particular mutation. In rare cases, the genetic analysis is inconclusive. In these cases, the genetic change will be further investigated in the laboratory. ENCORE contributed to the improvement of NF1 diagnostics (van Minkelen, Clinical Genetics, 2013).
Within ENCORE we also looked at the effect of the type NF1 mutation on cognitive functions (IQ). We found that all different NF1 mutations produce similar effects, with the exception of the so-called chromosomal micro-deletion. NF1 patients with this mutation are more affected (Ottenhoff, Genetics in Medicine, 2020).
Detailed knowledge about the course of Neurofibromatosis type 1 (which symptoms and complaints are there, and when exactly do they arise) is of great importance in order to be able to recognize complaints early and treat them optimally in the future. In addition, this is of great importance for drug research (trials). After all, only if we can demonstrate that a new drug improves the quality of life compared to an untreated patient, will the drug actually be approved and reimbursed. ENCORE has done a lot of research in this area, particularly with regard to cognitive functions, and the impact that this has on daily functioning. ENCORE has also conducted extensive research into the mechanisms underlying the cognitive problems in both patients and mouse models of NF1 (see "Our publications" and the preclinical research pages on this site).
Clinical trials
ENCORE has conducted a number of clinical trials to investigate if the cognitive problems in NF1 can be ameliorated. We found no indication that statins improved cognitive function. It is currently being investigated whether Lamotrigine can provide an improvement.
The TRAIN study
In addition to these cognitive trials, clinical trials are currently underway with a "MEK inhibitor" that focus on the treatment of the plexiform neurofibromas that occur in NF1. Plexiform neurofibromas can be very damaging cosmetically, but can also lead to neurological deficit, bone growth, blindness, severe pain and aggressive cancer. They are difficult to operate because they have too good blood flow. That is why medicines are badly needed. Recent research shows that children with NF1 and a non-operable plexiform neurofibroma benefited from medicinal treatment. As a result of this study, Erasmus MC will conduct a study with trametinib in adult NF1 patients with plexiform neurofibromas that cause complaints. Click here for English information on trialregister.nl about this study (including inclusion and exclusion criteria).
Neurophysiological outcome measures
For conducting cognitive trials, it is very important to be able to determine quickly and objectively in a small group of patients whether the drug may work or not. That is why we are now focusing on developing techniques to determine these "outcome measures". We do this for instance by using transcranial magnetic stimulation (TMS).
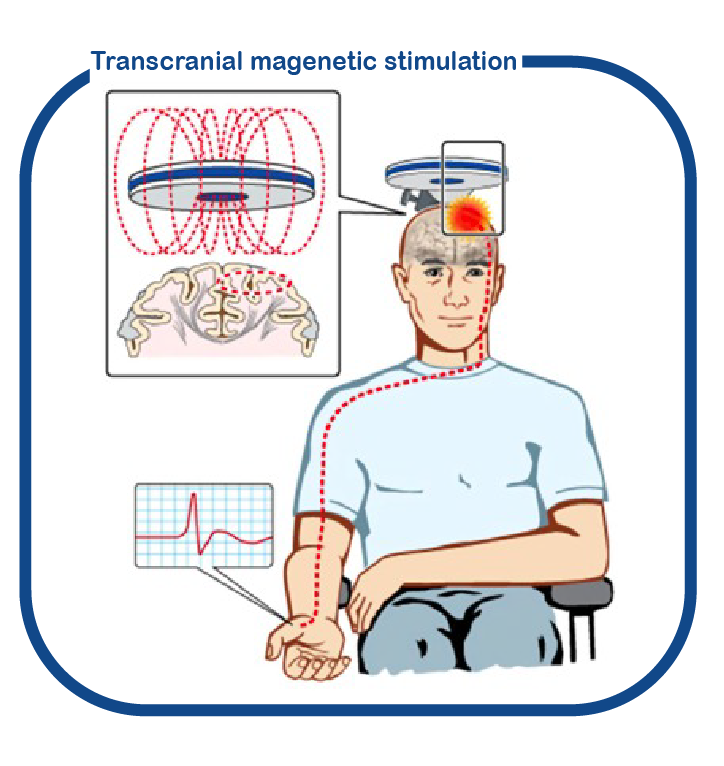
Bron: DOI: 10.3389/fneur.2018.01141
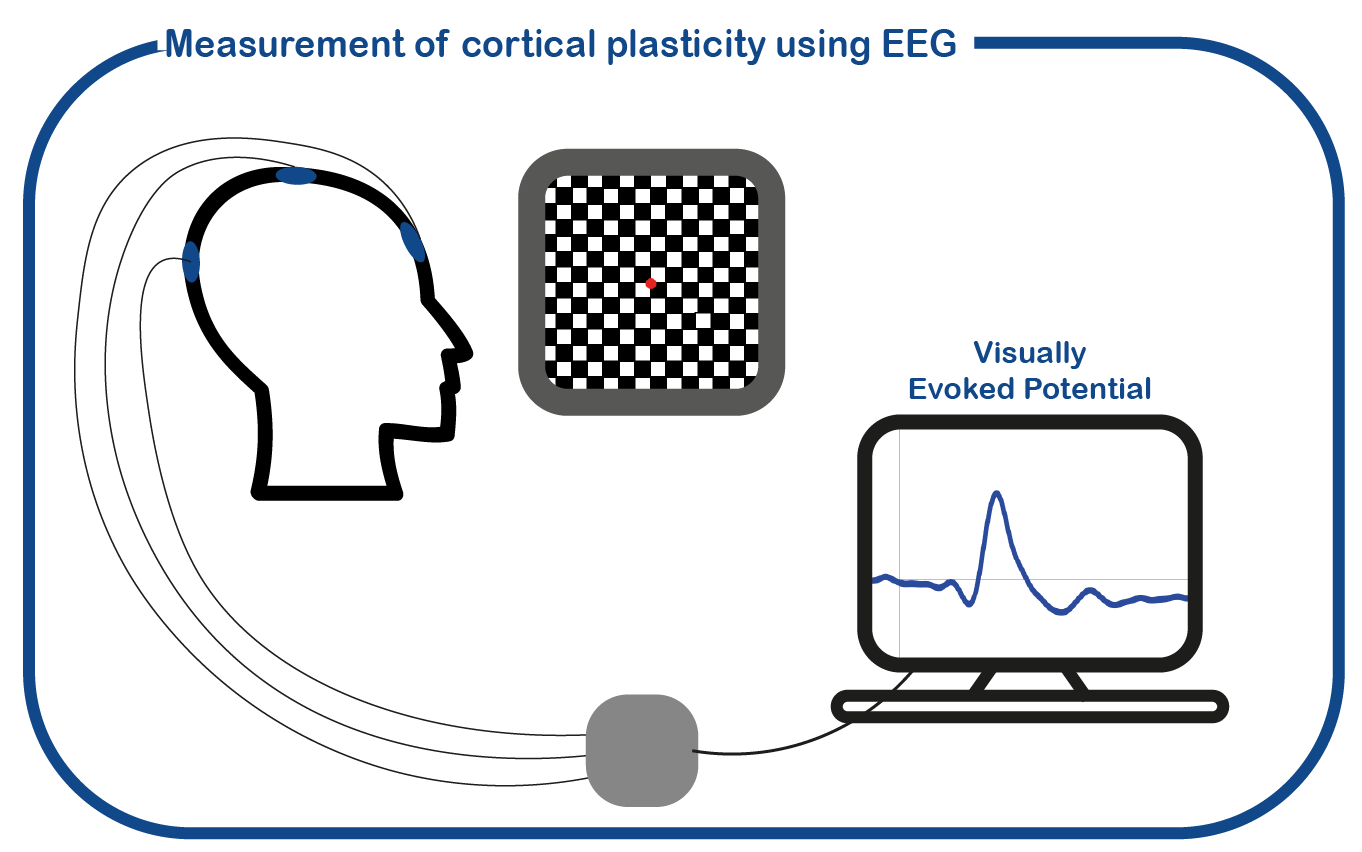
In addition, we use EEG measurements to measure the plasticity of the visual cortex by measuring "visual evoked potentials".
EU pearl
The ENCORE-NF1 expertise center participates in the consortium EU-pearl,, a collaboration between 8 hospitals, a number of pharmaceutical partners, and EFPIA, which aims to set up a framework for "multi-centre multinational platform trials" for a number of syndromes. ErasmusMC-NF1 is working group leader for "WP7 Neurofibromatosis". The overall goals are to establish a clinical research network for trials in NF in Europe, to develop platform trials for drug treatment of complications for NF, and to set up a long-term study of the history of NF manifestations. Click here for more information about the EU-pearl consortium.
FACE-NF1 - ‘MultiFACEt’ approach to predict the development of NF1
The complex course of NF1 unraveled from different perspectives. Using clinical data, hereditary factors and biomarkers, we develop a personal risk profile for people with NF1-related plexiform neurofibromas for tailor-made treatment advice.
This research aims to predict how benign and malignant tumors develop in people with NF1 by combining clinical, genetic and biological information. This will help to improve treatments and follow-up in the future. The strength of the research lies in setting up a national system where all information about NF1 patients will be entered and stored (a database), including the storage of tissue material that will be secured after an intervention or operation (a biobank).
Ottenhoff MJ, et al. (2025). The effect of lamotrigine on cortical inhibition and plasticity in Neurofibromatosis type 1: Exploratory analysis of a randomized controlled trial (NF1-EXCEL). Neurophysiol., Pubmed
Dhaenens BAE, et al. (2025). Identifying patients with neurofibromatosis type 1 related optic pathway glioma using the OMOP CDM. J. Med. Genet., Pubmed
de Brons B, et al. (2025). Identification of the Determinants of Plexiform Neurofibroma Morbidity in Pediatric and Young Adult Neurofibromatosis Type 1 Patients: A Pilot Multivariate Approach. Cancers, Pubmed
Lubbers K, et al. (2024). Autism Spectrum Disorder Symptom Profiles in Fragile X Syndrome, Angelman Syndrome, Tuberous Sclerosis Complex and Neurofibromatosis Type 1. Autism Dev. Disord., Pubmed
Ottenhoff MJ, et al. (2024) Lamotrigine for cognitive deficits associated with neurofibromatosis type 1: A phase II randomized placebo-controlled trial. Dev Med Child Neurol. Pubmed
Dhaenens BAE, et al. (2024) Platform trial design for neurofibromatosis type 1, NF2-related schwannomatosis and non-NF2-related schwannomatosis: A potential model for rare diseases. Neurooncol Pract. Pubmed
Dhaenens BAE, et al. (2024) Quality of life in individuals with neurofibromatosis type 1 associated cutaneous neurofibromas: validation of the Dutch cNF-Skindex. J Patient Rep Outcomes. Pubmed
Rosenberg AGW, et al. (2024) Endocrine and non-endocrine causes of fatigue in adults with Neurofibromatosis type 1. Front Endocrinol (Lausanne). Pubmed
Dhaenens BAE, et.al. (2024) The PlexiQoL, a patient-reported outcome measure on quality of life in neurofibromatosis type 1-associated plexiform neurofibroma: translation, cultural adaptation and validation into the Dutch language for the Netherlands. J Patient Rep Outcomes. Pubmed
Bennebroek CA, et.al. (2023) Treatment evaluation by volumetric segmentation in pediatric optic pathway glioma: evaluation of the effect of bevacizumab on intra-tumor components. J Neurooncol. Pubmed
Taal W, et.al. (2023) Symptomatische tumoren bij neurofibromatose type 1 Symptomatic tumors in neurofibromatosis type 1: a diagnostic challenge. Ned Tijdschr Geneeskd. Pubmed
Dhaenens BAE, et.al. (2023) Health-related quality of life of children with neurofibromatosis type 1: Analysis of proxy-rated PedsQL and CHQ questionnaires. Eur J Paediatr Neurol. Pubmed
Carton C, et.al. (2023) ERN GENTURIS NF1 Tumour Management Guideline Group. ERN GENTURIS tumour surveillance guidelines for individuals with neurofibromatosis type 1. Pubmed
Castricum J, et.al. (2023) Visual-spatial and visuomotor functioning in adults with neurofibromatosis type 1. J Intellect Disabil Res. Pubmed
Ottenhoff MJ, et.al. (2022) Cerebellum-dependent associative learning is not impaired in a mouse model of neurofibromatosis type 1. Sci Rep. Nov 9;12(1):19041. Pubmed
Douben HCW, et.al. (2022) High-yield identification of pathogenic NF1 variants in skin fibroblast transcriptome screening after apparently normal diagnostic DNA testing. Hum Mutat. Pubmed
Castricum J, et.al. (2022) Plasticity of visual evoked potentials in patients with neurofibromatosis type 1. Clin Neurophysiol. Pubmed
Lubbers K, et.al. (2022) Autism Symptoms in Children and Young Adults With Fragile X Syndrome, Angelman Syndrome, Tuberous Sclerosis Complex, and Neurofibromatosis Type 1: A Cross-Syndrome Comparison. Front Psychiatry. Pubmed
Dhaenens BAE, et.al. (2021) Lessons learned from drug trials in neurofibromatosis: A systematic review. Eur J Med Genet. 2021 Jul 5:104281. Pubmed
Castricum J, Tulen JHM, Taal W, Rietman AB, Elgersma Y (2021). Attention and Motor Learning in Adult Patients with Neurofibromatosis Type 1. J Atten Disord. Pubmed
Dhaenens BAE, et.al. (2021). Identifying challenges in neurofibromatosis: a modified Delphi procedure. Eur J Hum Genet. 26:1–9 Pubmed
Fangusaro J, et.al. (2020) Response assessment in paediatric low-grade glioma: recommendations from the Response Assessment in Pediatric Neuro-Oncology (RAPNO) working group. Lancet Oncol. 21(6); e305-16. Pubmed
Amirnasr A, et.al. (2020) Deregulated microRNAs in neurofibromatosis type 1 derived malignant peripheral nerve sheet tumors. Sci. Rep. 10:2927. Pubmed
Castricum J, et.al. (2020) Motor cortical excitability and plasticity in patients with neurofibromatosis type 1. Clin Neurophysiol. 131(11); 2673-81. Pubmed
Ottenhoff MJ, et al.(2020) Examination of the genetic factors underlying the cognitive variability associated with neurofibromatosis type 1. Genet Med.1-9. Pubmed
Frebourg T, et.al. (2020) Guidelines for the Li-Fraumeni and heritable TP53 related cancer syndromes. Eur J Hum Genet. 28(10); 1379-86. Pubmed
Siebelt M, et al. (2019) Congenital Forearm Pseudarthrosis, a Systematic Review for a Treatment Algorithm on a Rare Condition. J Pediatr Orthop. 40; e367-74. Pubmed
Vos JR, et.al. (2019) Boosting care and knowledge about hereditary cancer: European Reference Network on Genetic Tumour Risk Syndromes. Fam Cancer. 18(2); 281-4. Pubmed
Koczkowska M, et.al. (2019) Expanding the clinical phenotype of individuals with a 3-bp in-frame deletion of the NF1 gene (c.2970_2972del): an update of genotype-phenotype correlation. Genet. Med. 21(4); 867-76. Pubmed
Rietman AB, et al. (2018) Emotional and behavioral problems in children and adolescents with neurofibromatosis type 1. Am J Med Genet B Neuropsychiatr Genet. 177; 319–28. Pubmed
Rietman AB, et al. (2018) Worries and needs of adults and parents of adults with neurofibromatosis type 1. Am J Med Genet A. 176; 1150–60. Pubmed
Eijk S, et al. (2018) Autism Spectrum Disorder in an Unselected Cohort of Children with Neurofibromatosis Type 1 (NF1). J Autism Dev Disord. 48; 2278–85. Pubmed
Amirnasr A, et.al. (2017) Expression and inhibition of BRD4, EZH2 and TOP2A in neurofibromas and malignant peripheral nerve sheath tumors. PLoS One. 12(8); e0183155. Pubmed
Rietman AB, et al. (2017) Development of emotional and behavioral problems in neurofibromatosis type 1 during young childhood. Am J Med Genet A. 173; 2373–80. Pubmed
Rietman AB, et al. (2017) Motor problems in children with neurofibromatosis type 1. J Neurodev Disord. Pubmed
van der Vaart T, et al. (2016) Behavioral and cognitive outcomes for clinical trials in children with neurofibromatosis type 1. Neurology 86;154–60. Pubmed
Omrani A, et al. (2015) HCN channels are a novel therapeutic target for cognitive dysfunction in Neurofibromatosis type 1. Mol Psychiatry 20; 1311–21. Pubmed
Omrani A, et.al. (2015) Neurofibromin regulates HCN activity in Parvalbumin-positive interneurons. Mol Psychiatry. 20; 1263. Pubmed
Van Der Vaart, T. et al. (2013) Simvastatin for cognitive deficits and behavioural problems in patients with neurofibromatosis type 1 (NF1-SIMCODA): a randomised, placebo-controlled trial. Lancet Neurol 12, 1076–1083. Pubmed
van Minkelen, R. et al. (2013) A clinical and genetic overview of 18 years neurofibromatosis type 1 molecular diagnostics in the Netherlands. Clin Genet. Pubmed
Acosta, M.T. et al. (2012) The Learning Disabilities Network (LeaDNet): using neurofibromatosis type 1 (NF1) as a paradigm for translational research. Am J Med Genet A 158, 2225–2232. Pubmed
van der Vaart, T. et al. (2011) Motor deficits in neurofibromatosis type 1 mice: the role of the cerebellum. Genes Brain Behav 10, 404–409. Pubmed
Krab, L.C. et al. (2011) Motor learning in children with neurofibromatosis type I. Cerebellum 10, 14–21. Pubmed
Krab, L.C. et al. (2009) Health-related quality of life in children with neurofibromatosis type 1: contribution of demographic factors, disease-related factors, and behavior. J Pediatr 154, 420–5, 425.e1. Pubmed
Denayer, E. et al. (2008) Spred1 is required for synaptic plasticity and hippocampus-dependent learning. J Neurosci 28, 14443–14449. Pubmed
Cui, Y. et al. (2008) Neurofibromin regulation of ERK signaling modulates GABA release and learning. Cell 135, 549–560. Pubmed
Krab, L.C. et al. (2008) Impact of neurofibromatosis type 1 on school performance. J Child Neurol 23, 1002–1010. Pubmed
Krab, L.C. et al. (2008) Effect of simvastatin on cognitive functioning in children with neurofibromatosis type 1: a randomized controlled trial. JAMA 300, 287–294. Pubmed
Balgobind, B.V. et al. (2008) Leukemia-associated NF1 inactivation in patients with pediatric T-ALL and AML lacking evidence for neurofibromatosis. Blood 111, 4322–4328. Pubmed
van Engelen, S.J.P.M. et al. (2008) Quantitative differentiation between healthy and disordered brain matter in patients with neurofibromatosis type I using diffusion tensor imaging. AJNR Am J Neuroradiol 29, 816–822. Pubmed
Oostenbrink, R. et al. (2007) Parental reports of health-related quality of life in young children with neurofibromatosis type 1: influence of condition specific determinants. J Pediatr 151, 182–6– 186.e1–2. Pubmed
Cnossen, M.H. et al. (1998) Minor disease features in neurofibromatosis type 1 (NF1) and their possible value in diagnosis of NF1 in children. J Med Genet 35, 624–627. Pubmed
Cnossen, M.H. et al. (1998) A prospective 10 year follow up study of patients with neurofibromatosis type 1. Arch. Dis. Child. 78, 408–412. Pubmed
Cnossen, M.H. et al. (1997) Endocrinologic disorders and optic pathway gliomas in children with neurofibromatosis type 1. Pediatrics 100, 667–670. Pubmed
Cnossen, M.H. et al. (1997) Diagnostic delay in neurofibromatosis type 1. Eur J Pediatr 156, 482–487. Pubmed
Cnossen, M.H. et al. (1997) Deletions spanning the neurofibromatosis type 1 gene: implications for genotype-phenotype correlations in neurofibromatosis type 1? Hum Mutat 9, 458–464. Pubmed
Do you have questions about research at ENCORE? Or do you want to participate? Please contact us via encore@erasmusmc.nl.
